For this month we will follow up on the discussion of cleaning validation for tableting that was covered in the October Cleaning Memo by discussing cleaning validation issues for hard capsule filling equipment for solid drug products (powders, granules, and pellets). In January 2026 we will cover issues related to liquid filling of soft capsules.
In general, the concerns for capsule fillers are not as great as those for tableting equipment (see October Memo) for several possible reasons:
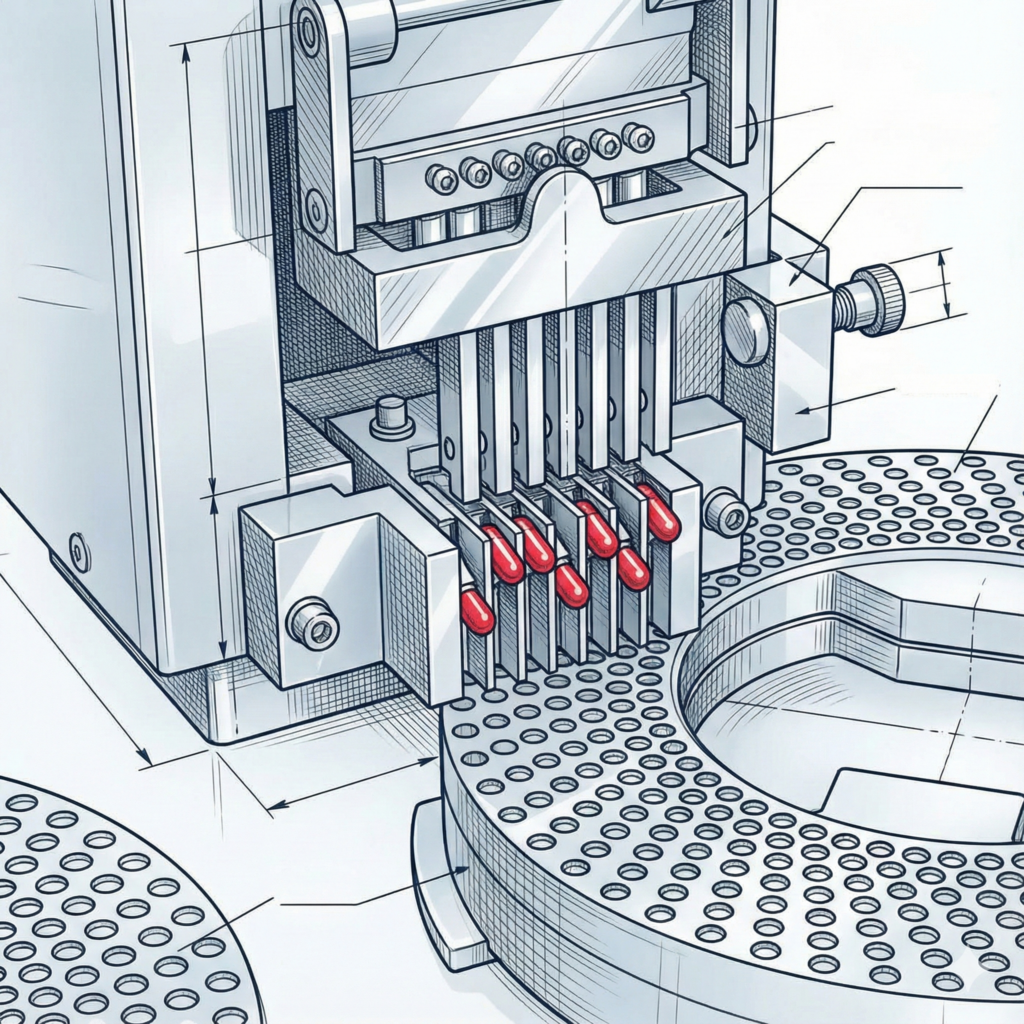
Let’s review what happens in a typical “dry product” hard capsule filler. Everything is contained in a polycarbonate chamber (to prevent dusts from escaping as well as to prevent dusts from the environment potentially contaminating processed capsules). The first step in processing is to place the capsules in the pocket or cavity in the lower ring. Then the top of the capsule is separated from the bottom by one of several mechanisms (such as vacuum assist or a straight mechanical one). So far there is no exposure to the drug product. Next is the is the mechanism for placing the solid drug product in the bottom segment in the proper amount and at the proper density. Mechanisms for powders include use of a dosing disc to fill a dosing chamber, which then using tamping pins to compact the powder so it can be dropped into the capsule lower body. Another mechanism is the dosater (dosing tube), which is more appropriate for granules/pellets because of better “flow” of these types of products. These are the equipment surfaces which have a more direct contact with the drug product. Once the drug product is in the lower capsule body, the upper cap of the capsules is lower onto the lower body and then pressure is applied to “lock” it into place. In some cases band sealing is done to further ensure the cap and body stay together (for example, as a “tamper evident” security feature). The filled capsules are then ejected for subsequent steps that might include polishers, metal detectors, and weight checkers before they are packaged.
Dosing devices for measuring product and placing it in lower capsule body
Unless these (the dosing disc/tamping pin or the dosater) are dedicated to one product, these are the most critical surfaces for cross-contamination of the prior product because they involve direct product contact. Typically these items are dedicated to one capsule size; the specific fill amount and fill density can be adjusted within a given range. So, if several drug products (using the same capsule size) are processed on the equipment, cross-contamination of a different drug active is certainly a possibility.
Parts (either individually or as a “block” assembly) are removed from the filler with cleaning done elsewhere. The cleaning process may be fully manual or in an ultrasonic tank. In either case, the cleaning process typically involves a preflush with water to remove loose product, a wash with a mildly alkaline detergent solution with multiple water rinses (WFI or PW). An additional alcohol rinse may also be used. The alcohol rinse helps to ensure that trace water is not left on the surfaces (although the parts are typically 316L stainless, so flash rusting in generally not encountered), as well as to reduce microorganisms on the cleaned parts. The cleaned parts are stored in a controlled manner to minimize external contamination before next use.
Other surfaces
This is where there may be a significant difference of opinion. How are surfaces like the upper and lower rings, as well as the lower ring cavities, the banding equipment and the polishers handled? My approach is to treat them as non-product contact surfaces. While these surfaces may contact the outside of the capsule, they typically do not contact the drug product inside the capsule. Furthermore, any residue transferred to the outside of the capsule will typically be the same product within the capsule. Is such transfer occurs, those residues should be significantly reduced by the polishing step. This assessment reinforces the needed to have effective cleaning of these types of surfaces with a requirement that they be visually clean.
Others may want to set more quantitative residue values. Unless a specific rationale can be made as to how to set such limits, those who want quantitative limits should consider having these part cleaned to the level as is done for “product contact” parts discussed below.
For such items removed, the cleaning process may be similar to the cleaning process for dosing items, or it may just involve a combination of brushing, vacuuming and/or wiping.
Non-product contact surfaces
Other filler chamber parts should be cleaned on a defined schedule. For clarification, product dusts may contact these surfaces, but generally those “dusts” are the same product being processed in the capsules (and are not a cross–contamination risk). So the key for cross-contamination is the changeover from one product to a different product. At changeover or within a campaign of the same product, cleaning processes are similar to the manual cleaning processes discussed above, involving some combination of brushing, vacuuming and/or wiping. Within a campaign the defined frequency will be established by the facility, and may be after a shift change or at a daily shutdown.
For such cleaning within a campaign, the concern is not so much cross-contamination but rather production efficiency and/or product quality. For example, do dusts build up such that capsules cannot be placed into or removed from the lower ring cavity, or such that fill weight amounts are affected.
Setting residue limits for product contact surfaces
This requires careful assessment. A key issue is to what extent the direct transfer of residues from equipment surfaces is such that the residue appears more or less uniformly in the processed batch. The worst-case scenario (resulting in lowest residue limit) is to include the surface area of those direct product contact surfaces in an overall (that is, cumulative) carryover residue limit for the processed batch. An alternative approach is that transfer of residues mainly occurs to an initial portion of capsules processed. If the latter is the case, then cross-contamination risks are significantly reduced since an initial number of capsules is typically discarded before capsules meeting QC specifications are consistently produced. This will depend on the facility and equipment (including the number of cavities/pockets in the lower ring) as well as the nature of the product processed. In calculating such transfer for the initially processed capsules, it must be recognized that for the dosing parts the “surface area to product weight” ratio will be much higher than similar ratios calculated for earlier process steps (such as granulation).
If you want to address the issue of preferential transfer by either establishing that it does not occur to any significant extent and/or to justify how much of an initially processed capsules in a batch should be discarded, the January 2025 Cleaning Memo has several options for types of this approach to evaluate cleaning limits at a product changeover.
For cleaning within a campaign, a formal validation study may not be required, but the cleaning should be documented at a minimum.
Note further that it is an expectation that product contact surfaces by visually clean after completion of the cleaning process (as well as at the beginning of capsule filling).
Testing for microorganisms may not be required if the product contact surfaces are given a final rinse with alcohol.
While this Memo focuses on limits for the capsule filling equipment, cumulative transfer of residues from all product contact surfaces must be considered to account for all sources of residues in the filled capsules.
Copyright © 2025 by Cleaning Validation Technologies