For this month we will follow up on the previous discussions of cleaning validation for tableting equipment (October 2025) and hard capsule filling equipment (December 2025) by discussing cleaning validation issues for gel capsule (also called soft capsule) filling equipment for liquid products.
For gel capsules, the situation is not unlike a form/fill/seal operation for small plastic vials. In general, the concerns for liquid gel capsule fillers are not as great as those for tableting equipment (see October Memo) for several possible reasons:
However, the concern for gel capsule filling may be greater because of the possibility of the liquid drug product drying on blending equipment and liquid drug product transfer equipment surfaces (and hence making cleaning more difficult).
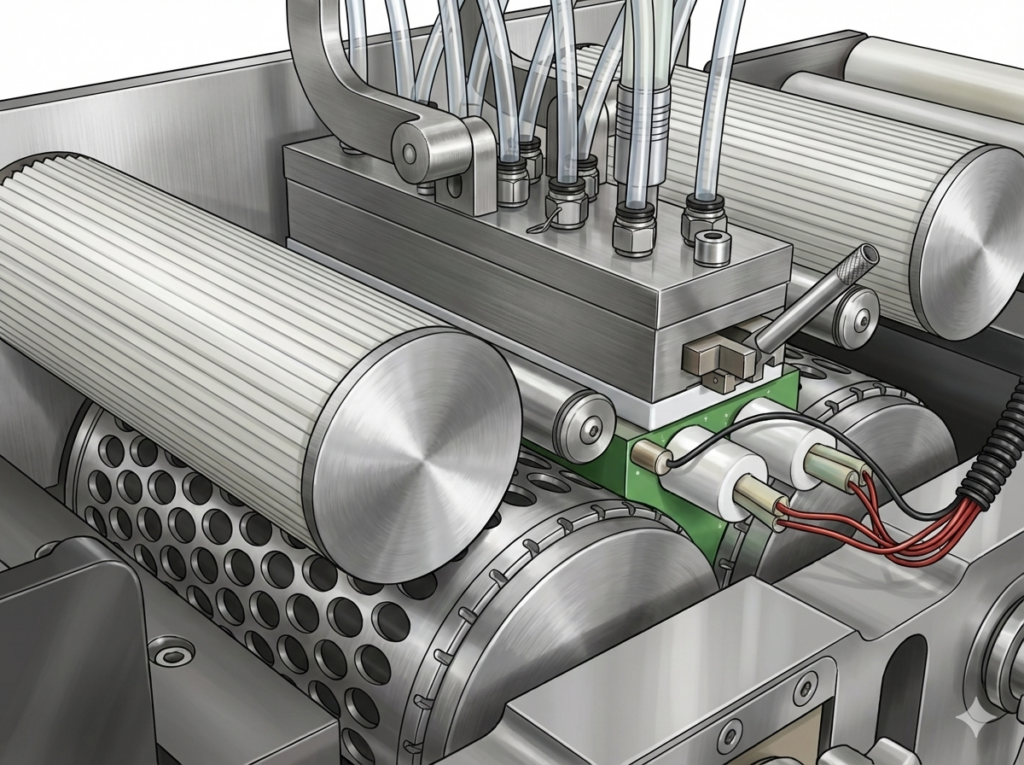
Let’s review what happens in a typical gel capsule filling processer. Unlike hard capsule fillers, the gel capsule equipment is generally not in an enclosed cabinet, but in room airspace with controlled temperature and humidity to avoid possible adverse effects on the gel processing. The liquid drug product, which is more typically in an oil formulation, is first blended so it is ready to be transferred to nozzles for metered dosing to each individual capsule. The gelatin formulation is blended/heated separately in another tank (or in separate tanks if the capsule is to be of two different colors). The gelatin formulation is then transferred onto the gel casting drums to form the gel “ribbons” (one ribbon for top and one for bottom). Each ribbon is then separated from its casting drum and moved to the die rolls. The top and bottom die rolls have matched cavities to “encapsulate” the liquid drug product.
Now is where the engineering “magic” occurs. More or less simultaneously, the two ribbons come together, drug product in metered between the two ribbons and the joined ribbons are sealed and cut to form individual capsules that are then discharged from the die rolls. The capsules are then dried in two steps. The first drying is tumble drying designed to prevent capsules from sticking together. This is followed by longer tray drying so the capsules assume their final shape and hardness. After inspection (to identify leaking, mis-shaped, or co-joined capsules), polishing (to remove any surface oils used as lubricants in various steps as well as to increase shine), and final QA, the capsules are ready to be released by QC.
Cleaning Issues
This discussion is separated into four areas based on the equipment used for those process steps: gelatin formulation (blending/heating tanks) and associated ribbon formation (drum rollers), drug product formulation (blending tanks), encapsulation (die rolls), and drying (tumble and tray dryers). Note that cleaning for the first type the primary concern is only cleaning of gelatin, while for the second type the primary concern is cleaning of the liquid drug product. The latter two types may involve cleaning both gelatin and drug product.
Unless drug product blenders, transfer hoses, and metering nozzles for liquid drug products are dedicated to one drug product formulation (as they might be for potent drugs), these are the most critical surfaces for cross-contamination of the prior product because they involve direct drug product contact. Because these typically are oil formulations, the cleaning process typically involves a wash with an alkaline detergent solution with multiple water rinses, followed by drying with a clean, lint-free cloth.
For gelatin formulations the blending/heating tanks, transfer hoses, and casting drums generally are dedicated to one formulation. However, if not properly cleaned and residues are left behind, there may be problems with the physical quality of gel ribbons processed (which is why campaign lengths may be limited to 48 to 72 hours).When switching from one gelatin formulation to a different one (such as with a color change), parts are more likely to be removed and cleaned “out of place”. The cleaning process may be fully manual or in an ultrasonic tank. In either case, the cleaning process typically involves scraping/brushing and/or a pre-flush with water to remove loose gelatin, a wash with a mild alkaline detergent solution with multiple water rinses (WFI or PW). An additional alcohol rinse may also be used help ensure that trace water is not left on the surfaces, as well as to reduce microorganisms on the cleaned parts (although that step may be unacceptable from an EHS perspective because the equipment is not generally enclosed). The parts are then dried with a clean, lint-free cloth. If cleaning is done within a campaign, such cleaning is generally done without disassembly.
For cleaning of capsule formation equipment (die rolls) within a campaign, parts are cleaned manually “in place” with brushing/scraping followed by wiping with a clean cloth and water (either individually or as a “block” assembly). For cleaning at changeover, parts are removed from the equipment with cleaning done elsewhere. The cleaning process may be fully manual or in an ultrasonic tank. In either case, the cleaning process typically involves a pre-flush with water to remove loose product, a wash with a mildly alkaline detergent solution with multiple water rinses (WFI or PW). An additional alcohol rinse may also be used. The alcohol rinse helps to ensure that trace water is not left on the surfaces as well as to reduce microorganisms on the cleaned parts. The cleaned parts are stored in a controlled manner to minimize external contamination before next use.
For discharge chutes and dryers, the main concern is leakage of drug product from an already formed capsule. Visual assessment of the equipment should be conducted, which may then lead to manual cleaning with an alkaline detergent and rinsing with water.
Setting residue limits for equipment product contact surfaces
This requires careful assessment. Certainly, drug product residues are of concern for the tank used for formulation of the drug product, as well as transfer equipment used to move it to the die rolls. Drug product residues may also be possible on the die rolls and subsequent processing equipment due to leakage during or subsequent to encapsulation. The concern for residues for the gelatin blending tank and the casting drums involves only the gelatin itself, and not the drug product.
A key issue is to what extent the direct transfer of residues from equipment surfaces is such that the residue appears more or less uniformly in the processed batch (as would be the case from residues in the drug product formulation tank). The worst-case scenario (resulting in lowest residue limit) is to include the surface area of those direct product contact surfaces in an overall (that is, cumulative) carryover residue limit for the processed batch. An alternative approach is that transfer of residues mainly occurs to an initial portion of capsules processed (as would be the case for the transfer hose and metering nozzles). If the latter is the case, then cross-contamination risks are significantly reduced since an initial number of capsules is typically discarded before gel capsules meeting QC specifications are consistently produced. This will depend on the facility and equipment (including the number of cavities in the die rolls) as well as the nature of the product processed.
For cleaning within a campaign, a formal validation study may not be required, but the cleaning should be documented at a minimum.
Note further that it is an expectation that product contact surfaces by visually clean after completion of any cleaning process.
Testing for microorganisms after cleaning may not be required if the product contact surfaces are given a final rinse with alcohol.
Copyright © 2026 by Cleaning Validation Technologies