For the last few Cleaning Memos we have deal with how ICH Q3A and ICH Q3B might be a help. Now we will try an approach that has been used in the past to deal with unknown peaks in HPLC chromatograms for measuring the target residue (typically the prior API or drug substance) in a cleaning validation protocol. This was discussed in the February 2005 Memo, although I had presented it in seminars/webinars after that date.
The approach has been twofold. One is to take preventive steps so that any peaks appearing in the protocol chromatogram can be identified so that they are not unknown peaks. The second is how to deal with peaks that are “truly” unknown (that is, those that cannot be accounted for by the preventive steps previously taken). We will deal with the preventive steps first.
Preventive Steps
This assumes that a HPLC procedure has been developed (and validated) for the residue of the prior API, so that the prior API can be measured on equipment surfaces at (and preferably below) any calculated limit.
So, the first attempt should be to identify both the cleaned product and the API of the prior product, and then develop a method for measuring that prior API in a pure solvent or solvent mixture (hopefully the solvent that would be present in the analytical sample to be taken in the CV protocol). The next step is to identify possible other residues that might be present in the CV sample to be analyzed by that HPLC method. Other possible residues that might show up as peaks in the specified HPLC method include: (1) excipients for finished drugs products (namely the cleaned drug product), (2) process aids, starting materials, or partial reactants for API manufacture, (3) cleaning agents, (4) cleaning process by-products, and (5) sampling materials (swabs, sampling solvents, sampling vials).It is not likely that excipients from a prior product would be present on equipment surfaces because those excipients would have to “survive” two cleaning processes, both the cleaning of the prior drug product and the cleaning of the target product (the only exception to this might be situations where there is external contamination by a prior product such as by airborne transfer).
I’ll illustrate how this can be done by determining if the cleaning agent appears or interferes in the HPLC chromatogram of the target API (that is, the API of the prior drug product). So we might perform several experiments. One is just to run the cleaning agent at its expected residue limit. The second is to perform the HPLC method with a sample of a mixture of the prior active (at its expected residue limit) and the cleaning agent (at its expected residue limit). The third might be to perform the HPLC method with a sample of a mixture of the prior active (at its expected residue limit) and the cleaning agent (at its expected residue limit), perhaps at the cleaning process parameters of time and temperature. Note that if the cleaning agent is highly alkaline or highly acidic, I might neutralize before analysis in order to avoid overcoming the buffering capacity of the mobile phase. There might be several outcomes, including:
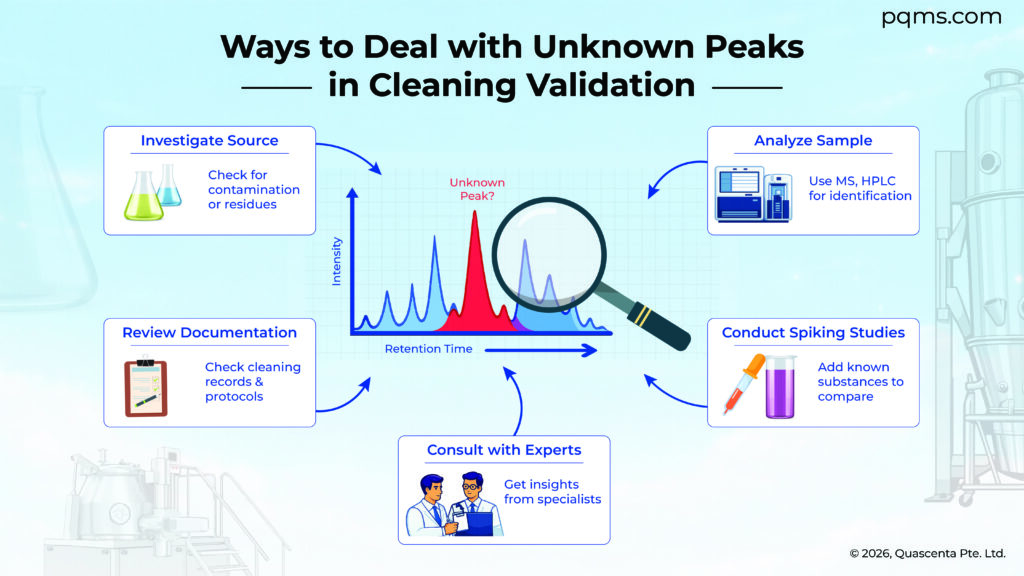
What is done in this illustration for the cleaning agent could also be done for other possible residues that might be present.
This preventive approach might be excessive work if there are no unexplained peaks in the chromatograms; in that case one of the following options might be used instead of this preventive approach. In either of these options, the cleaning process is performed, with samples collected and analyzed using the validated HPLC method for the target API residue and with the result that there are unknown peaks in the chromatogram.
Post-facto Option #1
In this approach an API at its residue limit in the sample being analyzed is spiked with each (or all) of the possibly interfering residues in an analogous way as described in the “preventive steps” above for the cleaning agent, and then the HPLC analysis is performed. If the API peak is as expected, any changes in the chromatogram may help identify the source or cause of any unknown peaks. It should be noted that if the spiking studies cause the disappearance of the API peak, that HPLC method might not be suitable for measuring API residues in CV protocols; the analytical group should be consulted for possible ways to deal with that situation. As in the preventive approach described above, consideration should be given to the effect of cleaning process temperature and time.
If the source or cause of any unknown peak is identified, then an assessment needs to done to see if it is present at an acceptable level, perhaps by utilizing the toxicity of that interfering substance. This could be done by use of data such as the PDE or ADE of the substance. Another option might be utilization of the approach covered in the March 2026 Cleaning Memo involving application of the ICH Q3B principles to determine whether the residues are below the specified thresholds.
Post-facto Option #2
In this approach the cleaning process is performed and the HPLC method for the prior API is performed as is typically done. The chromatogram is evaluated to capture any “unknown” peaks with its retention time and AUC (or peak height). Initially there are no spiking studies. For each unknown peak, the concentration or amount is calculated assuming that the detector response for each unknown is the same as the target API. That concentration (or amount) is compared to the concentration (or amount) of the target API at its residue limit, and then the percentage is determined. For clarification, this is not the concentration (or amount) of the target API actually measured.
Here is a clarifying example. Suppose the API is actually measured at 2, but the amount at the residue limit is 20 (meaning I would pass the protocol acceptance criterion for the API). Using the assumption of an equivalent detector response of the unknown substance, the concentration (or amount) of that unknown was found to be 1. The comparison I would make is “1 to 20”, or a percentage of 4%. For contrasting example, suppose the API is actually measured at 2, but the amount at the residue limit is 20 (again meaning I would pass the protocol acceptance criterion for the API). But again, using the assumption of an equivalent response of the unknown substance, the concentration (or amount) of that unknown was found to be 5. The comparison I would make is 5 to 20, or a percentage of 25%.
So what do I do with those percentages? What has been done in the past has been to specify a threshold percentage that should not be exceeded. For example the percentage values specified in that 2009 Memo are no more than 5% or 10% for any given unknown peak. I have seen some companies use a percentage as high as 20%. An additional criterion is to combine multiple unknown peaks in the same chromatogram, such that the sum of the percentages of all unknown peaks is no more than 20% or 40%. These conservative values are based on possible issues related to the relative response factor and the relative toxicity between the unknown substance and the target API residue.
If percentage thresholds are exceeded using this Option #2, then one should consider further evaluation using the approach in Option #1.
In any of these approaches it should be realized that what is appropriately done will depend on the specifics of the situation as given in high level polices or procedures.
The issues discussed above also may not be all the approaches to deal with unknown peaks. For example, some companies might decide to employ mass spec to help identify unknown peaks. Others may want to make their cleaning process more robust, such as by extending the rinse time to (possibly) entirely eliminate unknown peaks.
The point of this Cleaning Memo is to explore issues that might be addressed with “unknown” peaks in HPLC chromatograms in sample analyses in cleaning validation protocols. The point is not to proscribe nor prescribe certain practices, but rather to define some key issues and approaches.
© 2026, Cleaning Validation Technologies